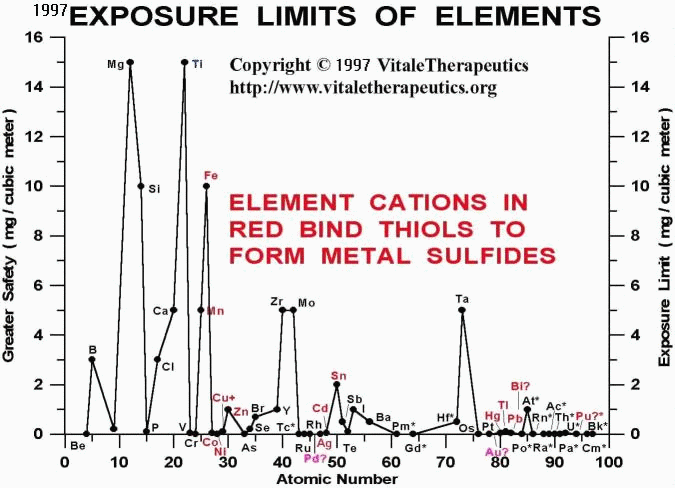

It is clear that cationic elements that bind sulfur compounds tightly (in particular those sulfur compounds with the aliases, thiol, sulfide, or sulfhydryl, RS-H) can be quite toxic. In the periods illustrated by highlighting, elements at the start of and end of each "period" are less toxic than elements in the middle of each "period". Thus, manganese (Mn@25), iron (Fe@26), zinc (Zn@30), and tin (Sn@50) are less toxic than copper (Cu@29), silver (Ag@47), cadmium (Cd@48), mercury (Hg@80), thallium (Tl@81), lead (Pb@82), and the components of stainless steel, cobalt (Co@27) and nickel (Ni@28), along with chromium (Cr@24). In the literature, there is a conspicuous lack of safety data for palladium (Pd@46), gold (Au@79), and bismuth (Bi@83). Note that two of these three are considered to be "precious" metals. With such little data on gold and palladium, it is a little surprising that we were able to find any data on the toxicity of another metal promoted as being "precious", platinum (Pt@78).
It is suspected that these supposedly "safe" precious metals and bismuth are anything but, and at least one toxicology text lists the same antidote (BAL or "British Anti-Lewisite") for the poisons, arsenic (As@33), antimony (Sb@51), mercury, bismuth, gold, nickel, and copper. BAL is also a thiol substance, 2,3-dimercapto-1-propanol, containing not one, but two sulfurs, and presumably should bind metal toxins more tightly than compounds having only one thiol (S) group, such as vitaletheine. Thus, BAL can be used as an antidote for these toxic metals probably because it binds the toxins more tightly than vitaletheine, thereby protecting this and other critical thiol substances occurring naturally in the body (glutathione, thioredoxin, lipoic acid, etc.) from chemical precipitation and essentially biological inactivation.
Arsenical compounds such as the chemical warfare agent, Lewisite, and arsenite, also are known to bind critical thiol compounds, like the vitaletheine modulators. Thus, bismuth and gold are recognized as toxic, with antidotes listed in toxicology texts. By association in the "periods", supra, the so-called "precious metals" palladium, silver, and platinum are also suspected of being quite toxic. With this information and in consideration of the discussion, below, concerning the use of bismuth salts for gastrointestinal upsets, neither gold injections into joints nor the wearing of copper bracelets to diminish the pain and inflammation of rheumatoid and other arthritic conditions can be advocated.
It is also significant that only 24 carat is considered to be "pure gold". Fourteen carat gold is "cut" with about 42% some other metals and 10 carat gold is about 58% some other metals. It is rumored among jewelers that gold is now being "cut" with carcinogenic nickel, itself sometimes referred to as "German silver". Gold has been routinely "cut" with toxic platinum and probably with toxic palladium for years. Together with the use of these toxic metals in dentistry and the use of silver in jewelry, dentistry, and orally in colloidal suspensions (as a supposed remedy for infectious ailments) these metals constitute a significant exposure of an affluent, industrialized society to unnecessary risks for serious, debilitating, and even fatal disease.
Gold, copper, and bismuth, though recognized as being toxic and
needing
antidotes, have been "promoted" as remedies for health problems for
years.
With this backdrop and based upon the emerging new information about
the
immune system, the use of these toxins, and even the use of colloidal
silver,
probably alleviates only the malaise associated with the immune
system's
inflammatory response against the pathological or parasitic organism
that
is actually causing the dis"ease". As such, these metals conceivably
serve
only as a temporary relief for the discomfort and actually may poison
the immune
system, thereby either prolonging the infectious part of the affliction
or weakening the immune system to such an extent that some other
opportunistic
infections can ensue, later. Both of the latter effects are "good
business"
for those hawking palliative and perhaps even counterproductive
treatments.
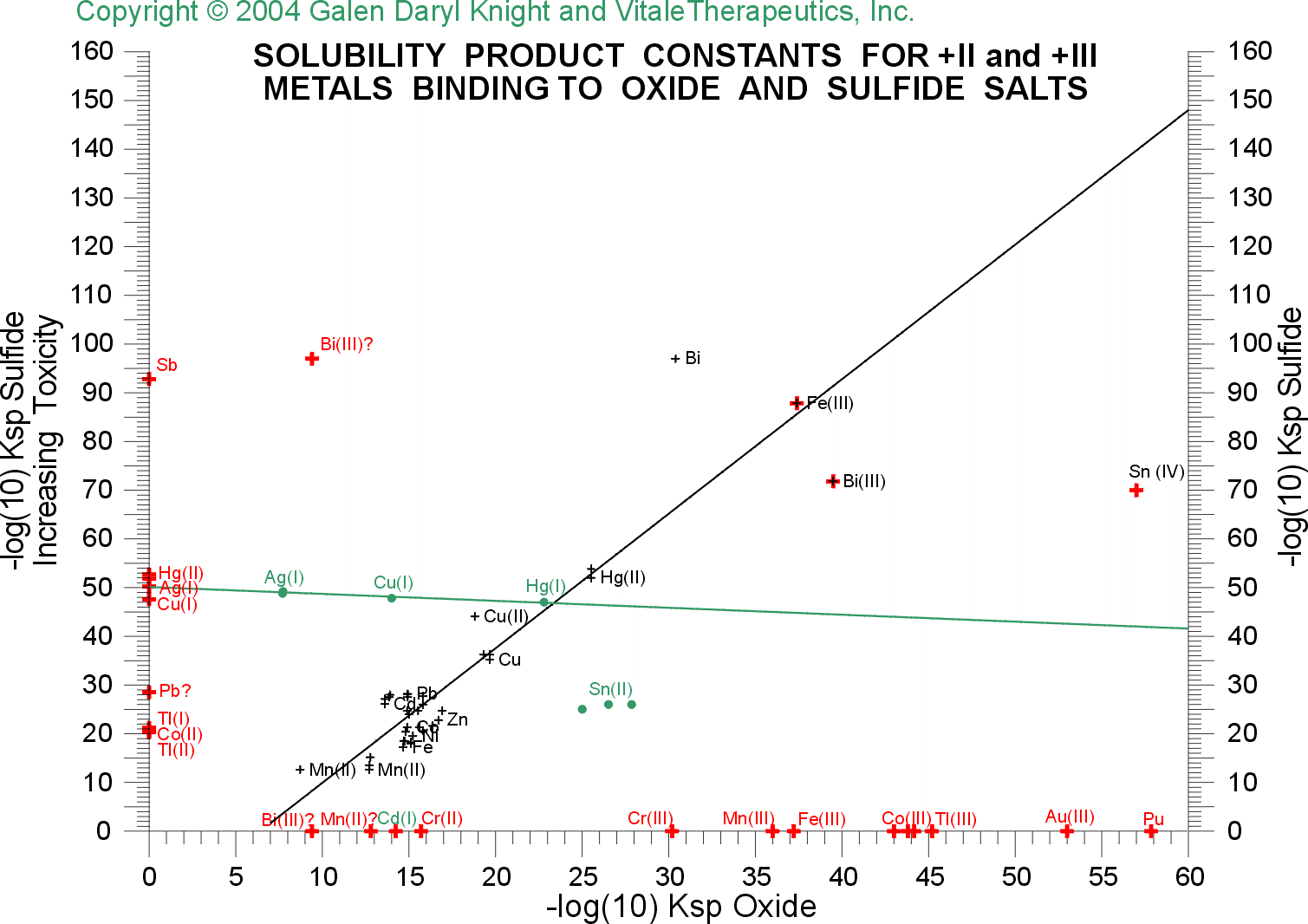
These concerns, and the ability of an extremely pure food grade
diatomaceous earth from Perma-Guard to
remove these toxic metals through its huge surface area of polymerized
silicate [from salicic acid, SiO(OH)2], are graphically
illustrated by simply comparing the solubility product constants of the
metal toxins with sulfur, as contained in the vulnerable
sulfur-containing compounds of the body, and correlating these with the
solubility product constants of comparable oxides and hydroxides from
water (or silicate), all of which are readily available from the
scientific literature and through the world-wide web:
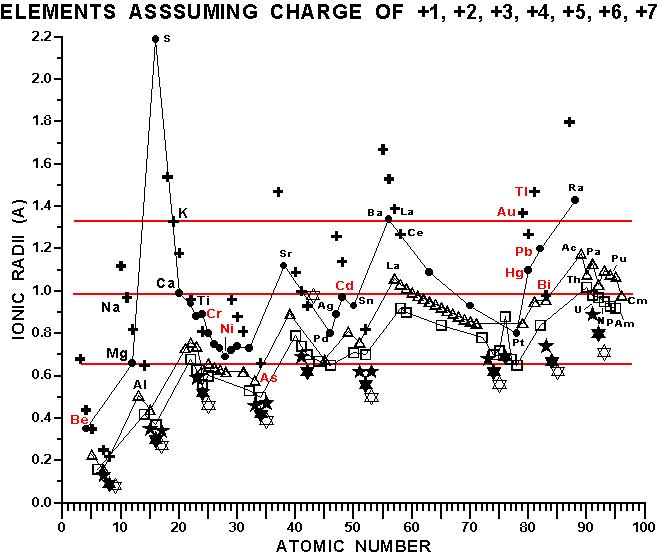
The following key identifies the oxidation state represented by each
symbol for each element in the figure, supra:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Several features of this figure are telling with respect to the chemistry and toxicity of these elements in our bodies. Sulfur (S@16) in our bodies in the +2 oxidation state has a very large ionic radii. This means that the electrons are held in shells a long way from the nucleus of the sulfur atom, making it more likely to be influenced by the chemical environment of the molecules that contain the oxidized sulfur. Because of this, sulfur in this oxidation state is known to be "highly polarizable", which translates into lay terms as being highly reactive, chemically. Thus, sulfur cations can be very good catalysts for chemical reactions, and our bodies make extensive use of this chemical fact.
The next interesting fact that emerges from this figure is that Beryllium (Be@4) is very small in its +1 and +2 oxidation states, which means that it can fit into the binding pockets on the receptor proteins and enzymes for many of the different nutritional cations in the body, thereby upsetting the natural balance in the body and causing a variety of health problems. Many DOE and DOD workers have been exposed to Beryllium and suffer a variety of ailments.
Aluminum (Al, atomic number 13) is also very small, so it can interfere with a variety of nutritional cations as well. Aluminum ions, together with nickel ions, can accumulate in the bone, replacing the magnesium (Mg, atomic number 12) that would otherwise build and maintain strong bones, especially when magnesium is deficient in the diet. Note that nickel (Ni, atomic number 28) and aluminum are slightly larger and smaller than magnesium, respectively. Substitution of nickel and aluminum for magnesium (because these ionic radii, when averaged, are not the exact same size as magnesium) can cause the atoms in the bone to fit together improperly and result in a weakening of the bone structures. This weakening of bone appears to be particularly problematic where the tendons and muscles articulate with the bone, resulting in stress fractures and tears that can be painful and debilitating.
Dr. Galen Knight was suffering from these very problems before he used the food grade diatomaceous earth to pull the nickel out of his body. As he pulled the nickel out of his body with the diatomaceous earth (please read disclaimer!), a painful condition where the tendon articulates on the outside of his left elbow simply went away. Nickel may not show up very well in a hair analysis because of its propensity to accumulate in the bone instead of magnesium, especially when magnesium is deficient, as it is generally thought to be in the average American diet. As expected from the similar ionic radii of nickel and magnesium, supra, there was not a great deal of nickel in Galen's hair analysis early in the DEtoxification program. However, upon supplementing his diet during the DEtox with a minerals containing magnesium, Galen noticed that his hair analysis started indicating much higher levels of nickel, and extremely high, even toxic, levels of aluminum. Thus, adequate magnesium supplementation may help drive nickel and aluminum out of the bones, strengthening them, and in the process making better use of calcium ions as well. Indeed, as Galen removed the nickel and aluminum from his system with the DEtox program, formerly high levels of calcium in his hair analysis gradually approached more normal levels.
These, albeit anecdotal, observations suggest that osteoporosis and other degenerative bone and joint diseases may be caused, in part, by accumulated poisons such as nickel and aluminum. This realization suggests a novel approach for preventing and even reversing these problems. For example, Galen went from not being able to use his left elbow, and having his left knee become very tender like a bad sprain during the year of DEtoxing with the diatomaceous earth, to being able to lift a 200 lb. person in his arms and do deep knee squats (not recommended!) holding the other persons entire weight.
Because hair analysis is not considered to be a good indicator of nickel toxicity, Galen also developed a blood test that appears to be somewhat specific for nickel poisoning. For example, individuals showing mercury poisoning in hair analyses do not give the characteristic toxic profile found in persons known to be poisoned with nickel. Clotting of healthy blood under carefully defined conditions normally entails an increase in viscosity, followed by a slight decrease, a second peak in viscosity, and finally a dramatic decrease in viscosity associated with a retraction of the clot that would normally pull the edges of a wound together to decrease the amount of new skin needed to heal the wound. A very sensitive instrument known as a Sonoclot is incredibly useful in following this process, and in detecting signs of cancer. Blood from cancer patients becomes very viscous in an amazingly short period of time relative to healthy blood, but this viscosity does not peak and retract like healthy blood. It literally goes off-scale on the Sonoclot and remains extremely viscous, without clotting and retracting. The nickel-poisoned individual displays a similar clotting profile, there is no peaking and retracting. The only apparent difference is that the viscosity builds much more slowly in the nickel-poisoned individual than in the cancer patient.
These observations may help to explain why nickel is known to be a contact carcinogen. By making the blood very viscous after an injury, but not allowing the normally short clotting period and retraction, the blood of nickel-poisoned individuals may become sluggish and unable to pass through the capillaries that supply oxygen to normal tissues. Warburg got the Nobel Prize for determining that cancer cells grow well without as much oxygen as normal cells. Other researchers have shown that when sulfur compounds are oxidized to disulfides, a process favored by oxygen in healthy tissue, the growth of both cancer cells and normal cells slows down. Thus, by thickening the blood and making it less efficient in transporting oxygen to healthy tissues, nickel-poisoning may create a condition that favors the growth of cancer cells. Galen's blood also displayed a stacking of red blood cells, known as rouleau, before he DEtoxed with diatomaceous earth. This stacking, like a roll of quarters, no doubt further increased the difficulty his blood had in passing through the tiny capillaries, thereby further decreasing the efficiency of his blood in carrying oxygen.
Nickel poisoning also over stimulates antibody production, as determined by Dr. David Lawrence, thereby increasing the risk of hypersensitivity and presumably autoimmune diseases, and confusing the immune system to such an extent that it may cause the immune system to miss cancer cells when they arise in the body. This phenomena is very interesting from the well known fact that cancer often "metastasizes" to proximal lymph nodes around the primary tumor. Consequently, cancer cells may be transported to lymph nodes to be killed by the immune system, but when the immune system is poisoned and confused to the point that it is attacking normal cells instead, the cancer cells can simply grow in the lymph nodes, unchecked. Galen is very suspicious that even primary tumors arise in lymph nodes. Thus, lymph nodes are not the enemy, and theoretically, the radical removal of all lymph nodes in the vicinity of the cancer, while also killing what immune system remaining with chemotherapy and radiation, may be a recipe for disaster. Such an approach to dealing with cancer can cause toxin-containing fluids (lymphedema) to accumulate in the vicinity of the primary tumor and in the extremities because there is no lymph system left to drain them away. This accumulation of toxic fluids poisons the immune system, further, thereby ensuring that it will be unable to deal with the traces of cancer cells left by the more conventional allopathic procedures.
This phenomena is very interesting with regards to additional observations made by Galen. When Galen was toxic with nickel and displaying the characteristic pinhead-sized blisters on the palms of his hands, he was "allergic" to aluminum-containing antiperspirants. Nickel is removed from the body by sweating and when this process is prevented under the arms by antiperspirants, the nickel probably accumulates in the skin and backs up into the lymphatic system. In fact, nickel is known to be a contact carcinogen and, consequently, can behave much like a carcinogenic tattoo, sweating out and reabsorbing. Hair loss, that Galen was experiencing when toxic with nickel, may also be a symptom of this toxic tattooing, sweaty brows, sweaty palms, and sweaty feet all being areas particularly affected by the nickel poisoning. It is also interesting that American Indians have long used sweat lodges for restoring health, Scandinavians have popularized saunas, and Americans used to use sweat boxes to get rid of toxins, such as nickel, to improve health and help overweight individuals metabolize fat away. When Galen started DEtoxing with the food grade diatomaceous earth, one of the first symptoms showing radical improvement was his "allergies" to antiperspirants. It was only after a year of detoxing that his 35 lbs. of middle-aged spread started melting away.
Can such insights from the figure, supra, be applied to other interactions between various ions in the periodic table? The answer appears to be a resounding yes. Dr. David Lawrence again provides additional keys to understanding this process. While nickel can over stimulate the production of cytolytic antibodies by immune cells, calcium ions appear to be absolutely essential for proper stimulation and regulation of this cancer fighting activity. Thus, cadmium (Cd@48) and bismuth (Bi@83), being almost identical in ionic size with calcium (Ca@20), theoretically can poison this part of the immune system. Cadmium is, indeed, carcinogenic, a finding also in keeping with its ability to bind and presumably inactivate the vitaletheine modulators. Bismuth is known for a biological activity that may be equally insidious.
Bismuth salts, the pink liquids and tablets available over the counter, are known to calm gastrointestinal (pepto- or peptic) upset, but lets look at what may be actually happening in this response. If one has a pathologic organism or parasite attacking one's alimentary tract, a healthy immune system will attack it and cause the affected tissues to become inflamed, swollen, or at least irritated and "upset". Bismuth salts, by interfering with calcium ions and by binding up vitalethine (both needed to stimulate antibody production), essentially shuts down this portion of the immune response, making the inflammation and discomfort go away. However, by poisoning the immune response, bismuth salts probably compromise the immune system's ability to properly address the infection, thereby allowing the parasite or pathogen to eat away at the alimentary tract, UNCHECKED. Leaky gut syndrome, food allergies, lupus and other autoimmune diseases, malabsorption syndromes, and even colon cancer are predictable results of chronic poisoning of the immune system in the alimentary tract with bismuth. Thus, cations the same size as calcium, as illustrated in the figure, supra, probably should be avoided as much as possible for optimal health. Note, for instance, that the transuranium and many other radioactive elements are about the same size as calcium, so it is not surprising that they are known to accumulate in the bone and marrow (sic., instead of calcium) where they destroy or cause cancer in the stem and other cells that make our immune systems.
This concept finds additional support in the allopathic recommendation that sodium (Na@11) be restricted in one's diet to prevent a variety of health problems. Evidence is emerging that ionic sodium may not be as much of a problem as the other elements, discussed supra, but moderation in the consumption of refined salt (NaCl) is probably prudent because of the possibility that too much ionic sodium in the blood stream can interfere with calcium metabolism, the two cations having very similar ionic radii. The problem with cutting salt completely out of one's diet is two fold. Sodium is involved in the maintenance of a variety of ionic pumps (such as in cells, sweat glands, and kidneys) that pump nutrients into the cells and waste products out of the cells and body, including toxins such as nickel. Ionic sodium also is involved in the maintenance of the proper amounts of fluids and blood pressure in the body, so the body requires a certain amount of sodium ions for optimal health.
The other problem with decreasing salt intake too much is that in land-locked areas of the world, iodized table salt may be one of the major if not the only dietary source of iodine. The importance of iodine in the production of thyroxin, an incredibly important regulatory hormone in the body, is discussed elsewhere on this web site. Iodine metabolism probably also is intricately linked with the phenomenally potent vitaletheine modulators, that are thought to regulate virtually every biochemical pathway in the body to some extent through its monooxygenase receptor. Together vitalethine and its monooxygenase receptor are thought to help regulate the thiol to disulfide ratio within each cell of the body. Although it has been known for some time that the activation of thyroid hormone (thyroxin or T4) to triiodothyronine (T3) is critical to the control of many pathways and processes in the body, little is known about the by-product of this thiol-dependent activation reaction. The by-product is probably either a biologically active sulfenyl iodide or iodide, both of which could interact with the monooxygenase receptor for the vitaletheine modulators and with the reducing machinery in the cell (such as glutathione, glutathione reductase, thioredoxin, glutathione peroxidase, lipoic acid, etc.) in a very dramatic and telling fashion.
Thus, blindly recommending that someone "cut the salt out of one's diet" should not even be considered by responsible health practitioners, nor should health conscious individuals even heed this type of irresponsible advice. The incorporation of sea salts, kelp, non-toxic mineral supplements, and sea weeds into one's diet should help deal with any problems caused by too much salt in one's diet. These tend to help the body balance a variety of nutritional minerals in sea salt by supplementing with, and thereby diminishing the cravings for, magnesium, calcium, zinc, and other salts thought to be deficient in the American diet. If one is not getting the required amounts of these and other nutritious minerals, a craving for salt will be the result. If the highly refined table salt (NaCl), devoid of these other nutrients, is the only source of salt consumed, over consumption of the less nutritious salt and associated health problems, such as interference with calcium metabolism (supra), will be the end result.
Two other glaring examples of conflicts between nutritious and toxic elements are evident in a comparison of ionic radii. Potassium (K@19) is a critical nutrient for the body. If the levels of potassium in the blood double from, or fall to half of, normal blood values, death can be the result. Muscle cramping is an example of one symptom caused by a potassium deficiency. When carried to the extreme of deficiency or excess heart attack can result. It is interesting that barium ions (Ba@56) are the same size as potassium ions and that barium ions cause a depletion of potassium ions from the body. Thus, "the GI series" in which one must first fast and then drink a chalky liquid so that the allopathic practitioner can visualize one's alimentary tract, may deplete potassium in the body. It may be a good idea to eat a few bananas or other good sources of potassium just before beginning the required fast. Caffeine, alcohol, and other diuretics, cortisone, sugar, and stress can also tend to deplete potassium, so these should be avoided if potassium homeostasis is a problem, especially during the fast just before a GI series. The cramping of muscles from vigorous exercise is another warning sign that potassium is being depleted.
GO TO:
| Home | Overview | People | Journal | Nutrition |
| Environment |
|
WWW Links | Outline | e-mail us |